Just like the Big Data (or Big Data Analytics), Translational Medicine has been another hot trend in recent times. Translational Medicine (TM) is also known as Translational Science (Research), Evidence Based Research, or Disease Targeted Research.
What is Translational Medicine?
Translational Medicine is the process of using the findings of laboratory research to develop new diagnostic tools and treatments, and similarly using the research findings from clinic (clinical trials) to develop new research ideas for laboratory research. Translational Medicine is based on three pillars – benchside (laboratory), bedside (clinic/hospital) and community (patient and general public involvement). It is kind of the interface between basic science and clinical medicine. The translation is not unidirectional. Translational happens while applying the knowledge gained through fundamental research in the lab towards new ways to diagnose and treat diseases. It also happens when clinical observations lead to new research issues (questions and ideas). So, bench to bedside, and back to bench. For in-depth understanding, please refer to Translational Research – Defining the “Ts” and T-Phases of Translational Health Research.
TM is extremely inter-disciplinary and a rapidly growing field in Biomedical Science. TM do not only brings specialists from various biomedical disciplines like biology, medicine, molecular biology, biochemistry, bio-statistics, bioinformatics; but also from chemistry (organic and analytical), physics and engineering. Yes, TM is as interdisciplinary as Biomedical Sciences or Pharmacy and Pharmaceutical Sciences.
The objective of TM is to combine the knowledge, resources, technology and applications of three pillars to facilitate the discovery of new diagnostic tools and therapeutic approaches using bench-to-bedside (B2B) approach through cross-functional collaborations between research scientists and clinicians, and using novel techniques and data analysis.
Different Phases of Translational Medicine
Basic Research
The basic research involves investigating fundamental science of human biology and pathobiology. People involved in basic science usually conduct research in the lab (bench work), which lead to identification of disease causing factors, drug targets (proteins or genes) and regulatory pathways (at genetic, molecular or cellular levels).
Pre-Clinical Research
This part acts as a bridge between the basic science and clinical medicine. During this stage, scientists take forward the findings from the basic science (laboratory findings) to further understand the aetiology of the disease, and also work upon finding treatment. Whereas basic science is (or at least should be) non-targeted; pre-clinical research is always targeted towards a particular disease. Scientists test hypothesis using cells, tissues, animal models. Use of various computer-aided simulations is also very common in order to facilitate the development and preliminary testing of drugs and diagnostic tools (or devices).
Clinical Research
Clinical research usually involves clinical trials of drugs in humans (patients and healthy volunteers) in academic medical centers and hospitals. The main objective is to test the safety and efficacy of the drugs (or diagnostic approaches); and to find out a correlation between a particular biomarker (disease marker) and patient phenotype (specific group of patients). One of the critical aim of clinical research is to obtain regulatory approval for a new therapeutic or diagnostic intervention.
Clinical Implementation
The next stage is the clinical implementations that primarily involve new interventions becoming routine clinical care for the larger population, and conducting research to evaluate the findings from the clinical trials. This is one of the major stages that leads to new clinical questions for the basic science.
Public Health
The final stage of translation involves evaluation of the health outcomes at the population level in order to determine the effectiveness of the new interventions for prevention, diagnosis and treatment of the disease. Findings may lead to working on improvements or development of new interventions.
What is the Need of Translational Medicine (Research)?
Whereas the traditional science has been more about strengthening the existing hypotheses, TM is all about creating insights into clinically significant interactions among drugs, pathways, targets and diseases.
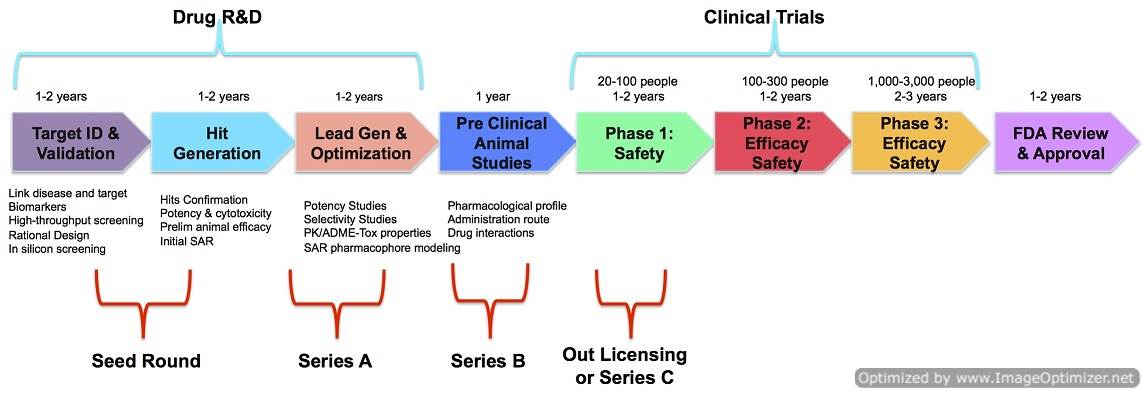
Image Courtesy: steveblank.com
As you can see from the above image, the drug discovery process is a very long (time consuming) and expensive process. Approximately, it takes around 15 years and USD 800 Million to 3 Billion to launch a new drug molecule. The R&D process starts with around 10,000 compounds, out of which 5 compounds end up in the pre-clinical phase and finally the world gets 1 successful drug molecule.
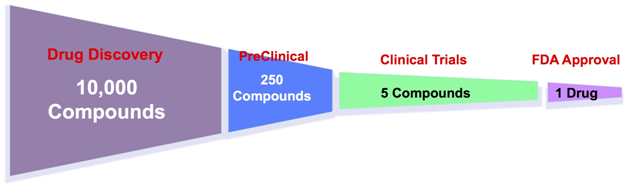
Image Courtesy: steveblank.com
Pharmaceutical companies, venture capitals and investors need to go through huge risk when it comes to spending on drug discovery. 75% of that money gets spent on the clinical trials and regulatory approvals. Even if a drug discovery project fails before the clinical trials (within the Drug R&D phase), investors might face a loss of around USD 100 – 250 Million. Whereas if the drug fails in any of the stages within the clinical phase, that will be a big ouch moment for the investors. Even the FDA evaluates drugs by their cost-effectiveness, so the end of the funnel needs to be large enough to justify the venture investment as well.
Before 1920, diabetes used to be an almost death case. Hence, insulin has been one of the most major triumphs in the history of medicine. The discovery and development of insulin therapy spanned over 150 years and that is the drawback of classical approach. However, it should be remembered that back in those days, the knowledge and technology were not that advanced.
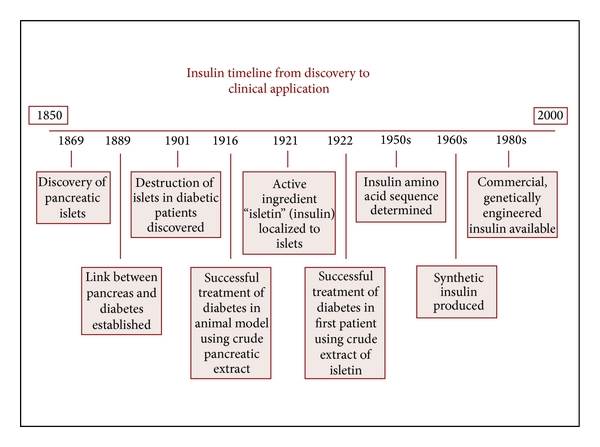
Image Courtesy: http://www.hindawi.com/journals/ab/2014/278789/
Eli Lily launched Humulin, an intermediate acting the first product of recombinant-DNA technology; the advancements are still in process. Fluorolog (by Thermalin), an ultra-concentrated fast-acting insulin formulation entered clinical trials in 2014. There should be more success stories like Lipitor, one of the all-time blockbuster drugs that generated a revenue of USD 13 per year for almost a decade. For curious folks, here is What Really Drove Lipitor’s Success. It is tough to re-create a success story like that of Lipitor’s as Matthew Harper wrote Why There Will Never Be Another Drug Like Lipitor on Forbes, but there is certainly hope for creating cost-effective and fast discoveries through TM.
Objectives of Translational Medicine (Research)
– To get the research from lab to benchside cheaper and faster; i.e.- accelerate the time process for bringing the intervention (drug. device or biomarker) from lab to clinic; and give the investors enough reasons to cheer about and keep investing at early stages.
– Create cross-functional collaborations among personnel working in different phases (basic science and clinic).
– Convergence of knowledge, technology and expertise from various disciplines and apply them to a common and targeted goal.
– To produce perfect solution for all patients, rather than just coming up with an okay solution for many patients. In a perfect world, hardly there is any drug that interacts with a single target that is linked to a particular disease.
Translational Medicine & Personalized Medicine
Whether the investment and efforts produces a result or not depends largely on genomic studies (sequencing) that can identify the set of patients most likely to respond to a new drug molecule. It would be unwise to expect a single drug treat all kinds of cancer. For example, Dabrafenib (GSK2118436) and Trametinib (GSK1120212) – two drug candidates from GlaxoSmithKline (GSK), reached the clinical phase with the aim of treating patients with advanced or metastatic melanoma with BRAF V600 mutation. Similarly, Roche and some other companies are also trying a similar approach to treat patients with BRAF V600 mutation. For interested folks – Role of BRAF V600 Mutation in Melanoma.
The advantage of translational medicine has been proved in both early phase and late phase drug discovery. TM provides feedback to pre-clinical scientists for further studies on an ad-hoc basis. During the final pre-approval stage TM provides insights into the target populations that may have a selectively better outcome and thus creating doors for personalized medicine. Hence the quest of finding new biomarkers is getting a lot of importance in order to stratify patient populations properly and provide a quantitative evidence of the benefits from the interventions. Modern translational medical researchers are even hoping to take the field to single patient level through stem cell technology in the coming future.
My First-Hand Experience in Translational Research
I have had quite a considerable exposure to different phases of translational medicine in four different labs across three different countries.
Master’s Thesis Project at Aston University (Molecular Pharmacology & Neuroscience)
During my MSc thesis at the Aston University, I worked on studying the mechanisms for the physiological actions of the CGRP protein. The CGRP protein is known to modulate various physiological functions and plays a major role in migraine and cardiovascular disorders. The CGRP protein exerts its actions through CGRP-Receptor (also known as CALCRL) and RAMP1 protein. My project was specifically on investigating the role of RAMP1 in receptor-protein binding and interactions between CGRP-receptor and Adrenomedullin. I was not working to find something new, but to re-confirm the hypothesis (reproducing previous findings are also considered as important as finding new results). The implications of receptor studies are always to lead to findings for new drug molecules. Without proper knowledge about how a receptor binds other molecules (or proteins), it is not possible to synthesize chemical compounds to target a specific receptor. I would term this study as basic science and the findings had the potential to lead to discovery of new medicines (or better medicines) to treat migraine (and many other disorders).
Graduate Research Assistant at University of Oxford (Cardiovascular Medicine, Molecular Cell Biology and Drug Discovery)
After finishing my Masters, I joined the Cardiovascular Division (Radcliffe Department of Medicine) at the Welcome Trust Centre for Human Genetics, University of Oxford. I worked on the protein called Creatine Transporter (CrT); and to be specific, myocardial CrT. For general audience – CrT is the protein through which creatine (supplies energy to cells in the human body) enters the cells. In case of low levels of creatine (Phospho-Creatine in correct scientific term) the heart cells starve of energy and that leads to myocardial infarction (heart failure or heart attack). My primary objective was to optimize a screening protocol and find chemical compounds (in collaboration with the Chemistry Department) to regulate the levels of myocardial creatine (in vitro), and any significant increase in the levels of myocardial creatine has got the potential to be beneficial in case of heart failure. This time I had a specific objective (targeted goal) while working on basic science in the lab; hence this was clearly a case of Pre-Clinical Research. During the project I did find few compounds that increase the cellular uptake of creatine and also identified few proteins that regulate the action of CrT. One of most important findings was the role of TXNIP for regulating CrT, and we also published a paper on that – A role for thioredoxin-interacting protein (Txnip) in cellular creatine homeostasis. In addition to that, the screening method that we optimized in order to identify new drugs also got patented.
Early Stage Researcher at Utrecht University (Cardiovascular Biology, Molecular Biology and Biomarker Discovery & Validation)
After a successful stint at Oxford, I moved to the Netherlands in search of new challenge and excitement. I was based at the University Medical Center Utrecht, and my project was discovery and novel biomarkers for prediction of cardiovascular events (heart attack, stroke, death etc.). I had the opportunity to work with human tissues (atherosclerotic plaque samples) and human samples (blood, plasma, and serum). I used to measure protein levels in healthy volunteers and patients and was trying to figure out the proteins that were elevated (or decreased) in patients in comparison to healthy people. After identifying the differential proteins, we were also used to study the basic biology and mechanisms of their actions, particularly of Osteopontin. This was something pre-clinical study and taking the findings from clinical studies back to lab bench.
Postgraduate Researcher at University of New South Wales (Cancer Biology)
The fascination for proteins and translational medicine took me to Down Under, where I took up another research position at the St. Vincent’s Centre for Applied Medical Research, affiliated to the University of New South Wales (UNSW) in Sydney. At UNSW, I was working on GDF-15 (also known as MIC-1), which is a biomarker for various types of cancer. It was also pre-clinical research with tremendous potential for translational medicine. For the first time, I gained hands-on experience on animal models (in vivo studies).
Requisite Education and Training for a Career in Translational Medicine
I had my Bachelor studies in Pharmacy (Pharmaceutical Sciences) and then moved to the United Kingdom to do my Masters in Pharmacology. It was a great learning curve as I gained exposure to different stages of Translational Medicine across various research areas. There is no hard and fast path for pursuing a career in translational research. Of course a Biology background is helpful, but you can also end up in TM with engineering background. Read the blog of Mechanical Engineering Graduate Ending Up in Translational Research. Most of the research these days is translational in nature. If you end up doing a PhD following basic or applied biological (or biomedical) science, you will have excellent scopes to conduct translational research. Recommended reading – Tips for Getting a PhD in Biomedical Sciences. Besides, there are few specialized Masters Programs that can give a perfect boost for a career in the translational medicine.
Top Masters Programs in Translational Medicine
USA
Joint Program – UC Berkeley and UC San Francisco
University of Pennsylvania
Temple University
Boston University
Medical College of Wisconsin
Canada
University of Alberta
UK
Europe
University of Helsinki
University College Dublin
Australia
Australian National University
Those who are interested in the field of Translational Medicine, should possess the “breadth”, as quoted by Robert Hertzberg of GSK. According to Hertzberg, a Physician or Medical Doctor should know about basic science and should have spent time at the bench in order to have a good and meaningful career in TM. Hertzberg further recommended moving around different parts of an organization instead of staying in a single niche. An ideal training would be to join an academic medical center (or clinical center) where you gets exposure to both basic scientific research and dealing with patients or clinical samples. A rounded experience backed by solid knowledge of basic and clinical science will be the key to career progression.
Scope of Translational Medicine (Research) in India
India has already been proved to be strategic location for conducting clinical trials by several multinational pharmaceutical companies. Besides, India is also rapidly getting in to a position for contributing towards drug discovery by adopting the translational framework. India has got a large pool both in terms of patients, and qualified and talented personnel. Other significant advantages are lower costs and English being the official language in the medical profession. Then there is the support from the Indian Government as well. Prime Minister Narendra Modi had declared USD 28 Billion spending on Universal Healthcare Scheme, which will provide free check-ups for everyone. This will provide an enormous clinical data. India has already got state-of-the-art data processing infrastructure for bioinformatics and biostatistics; and rapidly adopting the translational framework.
The set up of Translational Health Science and Technology Institute (THSTI), backed by the Ministry of Science and Technology (Govt. of India), in Gurgaon (2009) has been a critical step for revolutionizing translational research in India. THSTI has got a partnership with Harvard-MIT HST for infrastructure development and training. Courtesy to THSTI, the Drug Controller General of India (DCGI), Scientific Review Committees (SRC), the Indian Government, a healthy clinical trial infrastructure is getting set up all round the country. India is rapidly adopting a translational framework and all poised to narrow the gap between basic scientific research and clinical research as per the Critical Path Initiative of FDA. Hence, the prospects are quite bright for translational medicine in India.
We are NOT accepting new queries on this article. Instead, please post here – Career advice for Biotechnology careers
Very helpful as it gives very clear idea….thanks
Thanks, Chintal. I am glad that you found it helpful.
sir ur article is very informative thanku. now i finished my 12th nd i dont love chemistry but im ok with it but looking at broad oppurtunities in pharmaceauticals i want to persue it. but at d same time i love zoology so should i go to biomedical sciences (bsc) . plz let me know which has more scope nd my parents r not ready to send me abroad for studies as they r concerned of security issues .nd i want to go into research field.im totally confused nd also i am interested in veterinary too wat is d scope for that sir plz let me
Hi Ashwini,
You can definitely consider BSc in Biomedical Sciences or Biotechnology – you can gain a holistic overview about various branches. At Masters level, you can dodge Chemistry. However, you have to know the basic of Chemistry (especially Organic and Analytical Chemistry). At PG level, you can also consider MSc Genetics / MSc Biomedical Sciences / Veterinary or Animal Studies.
Tanmoy
Sir i am very happy to see the way you are so positively encouraging so many people and guiding them, i too need your valuable suggestions!! I am presently in third year of b pharmacy, i dont have much interest in chemistry but i am pretty sure i would be very fine with pharmacology and i aspire to do ms in pharmacology or either i want to make a career in mba but both of the degrees i would be doing from abroad itself.can u please guide me is it of any help for me to do gpat if i am very sure of going abroad? And about the 2 months training in summer holidays,can i do it in abroad? If yes,what procedure do i have to follow? I am good till now in academics and i have been the university topper throughout, which exams should i start preparing for? I would be very thankful to u if u answer my queries and guide me something.
Hi Aarti,
If you are clear that you want to do Masters from abroad then do go for it. However, preparing for GPAT side by side could be a good option even if you don’t want to study M.Pharm from India. The idea being that you will keep a competitive edge, and you will be brushing up your aptitude. It will be helpful in the long-term. Even I had made up my mind to go abroad when I was in the 3rd year (had started thinking about it in the 2nd year). But, I did attempt GATE (GPAT was not introduced back then) in both 3rd year and 4th year. The GATE preparation did help me to make my concepts clear and strong – those really helped me during my Masters from UK and later during my research roles.
It would be really nice if you do a summer training/project. Getting one in abroad is tough, especially for Indian students, but not impossible at all. I would suggest you to do a project from any of the Indian research institutes or IITs (IIT Kanpur, IISc etc.). In abroad you can look up EMBL or INSERM in Europe. In case there is any faculty members who has got some existing collaborations in abroad – these things do help in the real world.
Regarding exams – do prepare for GRE. Try to clear your GRE at least 15 months before the intake, and apply well before the application deadlines – that will give you advantage for getting generous scholarships.
All the best for your future.
Tanmoy
Thank you sir, again i would be very thankful i you guide me in detail about how to apply to embl?? I am very confused and being in a very remote area and the college i am studying in is not having any collaborations abroad!! [personal detail edited out] is my email address, i even have no knowledge about the project, is it that we can undertake a project in any year during our graduation?if u can email me the proper procedure or the link to apply for embl, i would be very thankful. Kindly help me.
Hi Aarti,
If you wish to apply to EMBL, check out the group leaders (http://www.ebi.ac.uk/about/people). You should then contact the group leader directly (via email). Projects also get advertised on their internships page, when available. You can undertake a project during any year of your graduation. Please be advised for summer internships, the application deadlines are usually Dec – Feb. But, keep checking for options, and be open to internships in India as well.
Few useful links:
http://www.goabroad.com/intern-abroad/search/biomedical-sciences/internships-abroad-1
http://www.peshaa.com/Internships/International/international-summer-internships.html
http://internshala.com/internships/summer%20research%20fellowship-internship
Best Wishes,
Tanmoy
Sir, I have completed my third of B.pharma and want to settle in Canada. I will be very obliged if you can guide me and answer following questions:-
1. Which is better option from PR and payscale point:- a. To take 1 or 2 year PG diplo course and give PEBC exam for becoming registered pharmacist. Or b. Do masters.
2. Is GRE mandatory for masters in Canada?
3. What are job opportunities if I go for masters in pharmaceutics?
Hi Anis,
Here are your answers:
1. Be it getting PR or Pay, I would advise you going after your interest. If you have got more inclination towards community pharmacy, you should go for PEBC exam. If you want to pursue a career in research or other functions – then a Masters will be better. If you acquire the specific skill-set, PR and good pay will not be a problem.
2. GRE is not mandatory for most of the Masters programs in Canada. However, if you wish to target top schools like UBC, Toronto or McGill, GRE will be preferred.
3. Pharmaceutics is a good option in terms of job prospects. You can go for Manufacturing/Production, QC/QA, F&D, R&D etc.
You can keep your options open by pursuing a Masters, and going for PEBC exam at the same time. Community Pharmacy has excellent job prospects in Canada. But, as mentioned earlier – do go consider your interests in the first hand.
Good Luck !
Tanmoy
I m in my final year bpharm in mumbai university and have cgpa of average 7.8 i want to apply for ms abroad in medicinal chemistry and molecular medicine plz suggest me good universities
Hi Hiral,
There are many good universities for MS in Medicinal Chemistry / Molecular Medicine. If you tell me your country preference, it will help me.
Country wise, Germany, UK, Sweden, US, Australia will be the top ones for Medicinal Chemistry and Molecular Medicine. You can also consider Singapore, Denmark, Poland and Ireland.
Hello sir it is really very informative to read ur posts n the queries and ur replies to them but I am a student and have cleared 12th boards with 78% and I always wanted to b a dermatologist..but as of now due to some reasons I was not able to attempt my state entrance exam gujcet and now I was planning for bsc but there’s a big confusion about which field should I go for that pays me well no matter what… bsc in biotechnology microbiology has a very less scope in india as said by few doctors I have met and also I m nt very much intrested in research work but I have heard of this bsc in food and nutrition to b an intresting field but I am scared n nt sure that is it really a well paying n good job offering field plzz help me out..I m damn confused abt what to do??!!
Hi Mahek,
It’s not true that Biotechnology or Microbiology has less scopes in India. But, it is true that the first 2 – 4 years in the biomedical field in India is not very well paid at most of the workplaces (academic or industry).
BSc Food Science and or Nutrition are nice options, especially if you’re not interested in research work but you have got a knack for biology. However, the 1 – 2 years might not be very highly paid everywhere. You can expect something around 15K per month. With 2 years experience and continuous skill (technical and soft skills) development, you can expect good career progress.
All The Best !
Hi sir,
I have done my bachelors in Biotechnology from Kolhapur, Maharashtra, India. now I’m Pursuing my final year in Masters in Stem Cells and Regenerative Medicine from Kolhapur itself. sir, as its my fina year i m looking for further careere opportunity. can you please guide me where i can apply for job opportunites in India??
Hi Priyanka,
You can have excellent job options in both academic and industry. In the academic domain, you can go for Project Assistant or JRF roles at research institutes like IISc (Bangalore), NCBS (Bangalore), MIRM (Bangalore), CCMB (Hyderabad) NCCS (Pune), TIFR (Mumbai), NIRRH (Mumbai), IIT Madras etc. In the industry, you can consider companies like Reliance Life Sciences, Biocon, Advinus etc. Try the job portals to get information for more companies (my personal recommendation – don’t apply through the job portals directly), and use LinkedIn to get connected with the right people (Talent Acquisition and HR Recruiters).
Refer to this article as well: http://www.thehindu.com/features/education/careers/career-options-in-stem-cell-research/article8085872.ece
Good Luck !!!
Sir,
I want to do B.Sc Biomedical Science from Delhi University. Can you suggest me after doing Bachelor in Biomedical Science, can i go for Masters in Biotechnology ?
I am doubt whether after b.sc medical science we can do masters in biotech or Not?
Help me in this as admission to DU is going to start and i want to know about the course B.sc Biomedical in details.
Hi Dhana,
You can definitely go for Biotechnology at Masters level after doing Bachelors in Biomedical Science. MSc Biotechnology will not be a problem at all. However, M.Tech. in Biotechnology at few universities might require a 4-year Bachelor degree – but that varies university to university.
Sir,
I just read your article about career options in biomedicine..but i did not understand your point about doing MD after bsc in biomedicine.
Could u please elaborate and suggest some colleges where students with bsc in biomedicine are admitted into MD course.
I have passed 12th std hsc exam from maharashtra board and confused about the openings.
Need your valuable suggestions.
THANKYOU
Hi Girija,
The fast-track MD program (4-year) is normally available in abroad universities. In India, you have to be a MBBS graduate in order to be eligible for MD program. In abroad (e.g. UK, Netherlands, Poland, Caribbean, Philippines) the first degree (undergraduate) program is a 6-year course. BSc graduates can get admission for the 4-year MD (graduate) program.
I hope this helps.
Thanx a lot for your help sir!
You’re welcome, Girija 🙂
hi im somdatta roy i hv completed my bphrm n qualified gpat now im pursuing mpharm in ceutics from Jadavpur University n meanwhile i hav done RA N PRODUCT MANAGEMENT courses from NCK PHARMA (online) wht n all i should do to upgrade myself. is sas training will be good enough or anything like that?after mpharm can i go for FND or marketing segment (not MR)?
Hi Somdatta,
After M.Pharm in Pharmaceutics you can go for F&D and R&D roles. In the marketing segment, you can go for PMT, but it would be difficult since PMT roles require 4 – 5 years of on-field sales experience and/or MBA degree. You can also consider Product Management or Brand Management roles (refer to this article – https://www.careerizma.com/blog/brand-management-careers-india/), but the entry could be a little but difficult unless you have got some real-world experience. One or two internship can help. Alternatively, you can start as Medical Representative, and work for at least a year. Get in touch with other experts in the PMT roles on social networking sites; LinkedIn is very much recommended.
Best Wishes,
Tanmoy
sir thanku for guidng me. sir vich is better in b pharma and docterate in pharmacy (pharma d -6 years course). vich gives me more exposure to research nd job oppurtunities at d same time
Hi Ashwini,
If you are interested in research work, then B.Pharma would be a better study option. Going for Masters (MSc or MRes) after Pharm.D., and later PhD is also possible. But, Pharm.D. will consume two years more than B.Pharma.
Hope this helps !
hi sir..i really need your help..
i passed from 12th class in 2014 (PCB) with best of four score %- 90.75%..
my dream was to be a doctor..i dropped first year..couldnt get into medicine..joined bsc life sci course in D.U. but then due to my depression condition i left that also..again prepared for pmts..this year there will be neet phase 2 in july and till then every admission will be over…
i want to build my career in a good field which is like medicine only if i m unable to crack medical entrance this year as well..
everyone thinks that i can do nothing in my life as i dropped two years..most of my friends will be in final year this year..
i am thinking of getting into bsc biomed..but in D.U. the cutoff for this specific course is high as 95%..
if not this then bsc biotech..but i m not finding good colleges for that..is a gov college important for ug degree??
and if i m able to get into this field then will it be a good career option as per pay scale and sucess rate ( in india and abroad)..
it will be great of you if u guide me..thank you
Hi Amy,
Your Class 12 score grade is excellent. It’s true that your 2 years got wasted, but you had a medical condition – and that’s absolutely fine. If you have got the passion and determination, do not worry about what others are saying.
First of all, I wish you all the best for the NEET phase 2 exam. In case you don’t make it to MBBS or BSc at DU – that’s not the end of the world. If you refer to my previous article(s), you will find list of good colleges in India. I also mentioned in one of my earlier posts that a good/reputed college does help, but the main thing will be what you learned and what skills you acquired.
Right now concentrate on the exams. We can discuss about the next steps later on.
Best Wishes,
Tanmoy
Hello sir,
I am very interested in pursuing a career in the field of chemistry , I have deep affinity for chemistry and i also love biology.Will B pharm be a good option for me? And what are the prier institutions in india for M pharm and pursuing a Phd in india and abroad. Can you please list out the entrances.And also give me some guidance as to how to prepare for gpat and niper so that i can concentrate from the very beginning itself. Please also tell the jobs in research field and are there any summer programs for B pharm students . Can the summer programs in iiser be attended by Bpharm students because they have mentioned BE/Btech/Bsc or equivalent degree as the eligibility.
Hi Rohang,
If you enjoy both Chemistry and Biology, then B.Pharma will be an excellent option.
Regarding GPAT preparation, just keep your basics (physiology, anatomy, organic and inorganic chemistry) clear from the very beginning. No need to start any grueling GPAT preparation from the 1st year itself; that can be delayed till the 4th semester. Start preparation after the end of the 4th semester, and attempt GPAT in both 3rd year and 4th year.
Summer programs do accept B.Pharm candidates. Apart from IISER, you can also try IIT Kanpur and other research institutes.
Regarding research jobs and other roles, please refer to my earlier article – https://www.careerizma.com/blog/pharmacy-careers-jobs/
Good Luck !
thanku for guidng me sir. how is zoology- biochemistry-microbiology combination? coz i thought it had more scope in research. i want to do bsc in this combination nd msc in biochem will there b more job oppurtunities in this field in india? if so vich r those?plz let me know!!!!!!!!!!!nd ihav got 94% in PCM &PCB nd interested mainly in zoology.
Hi Ashwini,
Zoology-Biochemistry-Microbiology would be a very nice combination for Bachelors. After MSc in Biochemistry, you go for research in the core Biochemistry, Chemical Biology or Systems Biology. Job prospects are good both in academia and industry. A PhD will be more helpful in terms of job prospects.
Sir i am 4 th year 8 th semester student from g.c.t.s asansol. My h.s mark is 62 only science, and my m.p marks is 66.and my b.pharm mark is 7.65 upto third year. I am attend gpat exam in 2016, but i am not qualify gpat exam . I am interested biology and chemistry But i am poor student for marks, sir what shall i do? Job or student .i am not interested in marketing job. Sir i am decided to enter hospital pharmacy job and get coacing for gpat 2017, sir i am qualify saat exam in orissa for m.pharm but someone telk me any pharmacy institute in orissa is fake so, i am not attend in councelling sir what shall i do? Please help me quickly and any good gpat coaching institute or gpat preparation tips please help me quickly.
Hi Souvik,
First of all, you have maintained 60% and above throughout your academic career; and that’s not at all poor.
In case you’re not too keen for marketing roles, and fail to crack GPAT (just in case) – you can think about QC/QA roles. Do try harder for GPAT next time.
You can get in to Hospital Pharmacy even with B.Pharm degree, though that will be super competitive.
There are quite a few colleges in every state in India which are very much below par (almost fake). But, there are decent colleges as well. You don’t need to listen to everyone. Speak to the right people, and do your own research as well.
I am not too sure about GPAT coaching centers, but you can try the following ones:
RAHE
Score tutorials
Resonance
The Gate Academy
ACE Engineering Academy
IMS
Pharmaglimps
Elite Academy
You can also try the online one: http://www.gpattutor.com/Purchase/gpatcourse.aspx
Hi sir,
I have appeared for MSc exam in biophysics. I am interested in research work in life sciences as well as radiation biophysics. Can you help me ?
Hi Dhanashri,
You can opt for research in the fields of Radiation Biology, Free Radical Biology, Radiation Therapy, Radiation Oncology, Medical Physics. You can go for Doctoral Studies in US, Australia and Europe.
I would also suggest to look at the International PhD program at FCT/UNL:
https://sites.fct.unl.pt/rabbit/home
Sir,
I am currently pursuing B.sc biotechnology from Kalyani University. I wish to do my masters in biotechnology from abroad. Is a good score in gre and toefl enough for getting a position in the top universities of usa?.i dont have any research projects,no internships under my belt. Should i only focus on grabbing a good score in gre and toefl. It will be very nice of you if you kindly tell me the path how should i proceed from today onwards in the aim of doing masters from abroad.
Hi Siddhartha,
If you’ve got a good academic track record (say more than 75 – 80%), an above average GRE score (310+) and around 95 in TOEFL, you can get into the top universities in the US (at least within top 75). Getting in to Top 20/40 could be a little tough without research projects and/or internships though.
Please be advised that few US universities do not accept 3-years Bachelors. But, there are many who do accept 3-year Bachelor degrees from India.
Apart from GRE & TOEFL scores, you will also need to work hard on your Personal Essay and Recommendations.
Hope this helps !
Hi Tanmoy sir,
I am NAZIFA, from india.I have just completed 12th n looking for career in Pharmacy but i m totally confuse in B.Pharm n Pharm.D….can u plzz help me taking my decision
Hi Nazifa,
I had answered a similar query earlier; please look at the above comments.
In short, if you’re interested in pharmaceutical industry and/or research, then B.Pharm is a better option. In case you want to pursue a career in community pharmacy, Pharm.D. will be ideal.
Hello Sir,
I am a B.Pharm student and I am going to be in final year. I am going to apply for 2017 fall. I wanna pursue a course that integrates genetics, pharmacology and chemistry. I am mainly looking for Universities in US. I have a decent gpa with internship experience. However, I am confused between Phd and MS. Can you please help me with this by stating the prospects of both the courses and What they demand with few suggestions on universities.
Thank You
Hi Anjana,
With a B.Pharm background I would suggest to go for MS first.
I am not aware of any specific program that contains Genetics, Pharmacology and Chemistry. But, biomedical science is very overlapping. So, if you go for MS in Molecular Medicine / Molecular Pharmacology & Genetics / Molecular Biology & Genetics / Biological Sciences / Chemical Biology, you can cover most of the key aspects. Later during PhD you can pursue those areas in more details.
Hope this helps !
Hi Sir, I have completed M.Sc in biotechnology from Pondicherry university. Then I worked as project JRF in research project related to cancer cell migration. I wanted to learn more about the human physiology and pathology so I looked for MS in biomedicine courses. I got admit from Iowa State University for 1 year program in Biomedicine. It is a non-thesis program but had a creative component for which I need to select faculty adviser and work on given topic. My future plan is to pursue PhD after the MS program preferably from USA or Europe. I just wanted to know how will the 1 year course effect on my PhD application? Also I wish to work in area of Biomarker for early cancer diagnosis as you mentioned in the article. Any suggestions and inputs as to how should I proceed further after MS would really be a great help. Thank you already!!
Hi Prita,
The 1-year MS in Biomedicine program at Iowa State University (ISU) will add value to your profile while applying for PhD programs, but only in terms of exposure and networking. If you network rigorously, it should be an excellent stepping stone for PhD (particularly in the US).
Personally I am not much in favor of doing two Master degrees, especially if they are in similar subjects. I prefer working as a RA or JRF after completing Masters, and then go for PhD. But, getting such a position in the US while applying from India is not too much feasible. From that perspective the 1-year MS program is a good idea, as it will give you a push for landing up with a PhD offer at a good US school.
Be focused from the very beginning during the MS program. I am not sure about the Creative component (what it actually is). As long as you’re getting some research exposure, it should be fine. But, if it is solely a theoretical program, then it could be a waste of time and money (unless you’re getting a generous scholarship or sponsorship).
Get in touch with your supervisor, and discuss about your research interests (biomarkers for early diagnosis of cancer). If you end up with a good research project (it’s fine if you do not write a thesis), conduct some nice experiments – that would be good enough.
Entry criteria for PhD in US and Europe are different. If you do your MS in Biomedicine from ISU, I think US will be an easier destination for you. But, that does not mean that you cannot get a PhD in Europe.
I hope I am clear enough. Feel free to write again if you need further insights.
Hi sir,
In which prospects is a PhD degree in stem cell and regenerative medicine beneficial over an Msc? Does it indicate to get a high salary package after completing a PhD? My personal choice is joining the industry not the academics so which will pay me better an m.sc degree or a phd degree?
Hi Navoneel,
In terns of pay, of course a PhD degree will give you a higher package. But, if you are not too keen about research (PhD in biomedical sciences will require a lot of energy and patience), it’s better to stop after MSc only.
However, a senior research position in industry will demand a PhD degree. But, if you want to get in to other roles – sales, business development, then MSc (or MSc + MBA) will be a better career path.
hello sir,highly impressed from your pharma article …basically i want to know after the completion of b pharma what should i do? mba or m pharma
Priyanshi, it completely depends on your interests. Both M.Pharm and MBA has got bright prospects. You need to choose the one that is better suited for your profile and career goals.
hello,I m a msc hons(microbiology) passed out student from panjab university last september. I m looking for a job. I m gate qualified. Not interested in phd,what is the scope of mba in healthcare management and its future prospects?Also any related guidance connected with my field??
Hi Shivani,
MBA in Healthcare Management will be a good choice with a Microbiology background. In India, hospital management and hospital administration are quite in demand for candidates with proper skills and knowledge. With growing importance on quality of healthcare and patient satisfaction (or customer satisfaction you can say), there is indeed a need for qualified persons in the field of healthcare management.
In the initial years, working hours could be a problem, but the compensations are good (not only salary wise, but also other benefits). There are rooms for career progression (e.g. Hospital Director). Besides, academic roles (like Asst. Professor) are also there to grab.
Hi I am Venkat completed MD Biochemistry from Dr NTR university Andhra pradesh. Please advise me, career options abroad.
@Venkat – are you looking for clinical roles, or research roles? If you want to get a mix of both, then above post is there for you.
hi tanmoy sir,
i am pursuing my b. pharm degree (III YR ) and my college is providing with summer internships so i am a bit confused about weather i should choose a low level pharma company or a globally established companies like glanmark etc. because some peoples are saying that these big companies do not allow students to practice inside and only provide us with certificate only without doing any work .
kopran mumbai was offeres to me but i refused .
so pls suggest me something
Hi Akash,
Apologies for the late response !
You have asked a very good question. To be honest both scenarios have got their pros and cons. Undergoing a training in a small company will give you a good exposure. You will find out how things work out in the real world. However, that will help you only if you are gong to end up in the Production, QC or QA roles. Besides, I assume you will be undergoing the training for 2 – 4 weeks (which is quite common for all Pharmacy students). So, that’s not a very long time.
On the other hand you might not get a very good exposure in a big company straightaway, but if you approach in a proactive manner, that can be possible. Moreover, if you are social enough, you can also use the opportunity to build some good contacts – that will help you in the long run.
I have seen my friends, batch-mates doing their internship/training from big companies like Lupin, Cadilla etc. They did gain some valuable experience. The best thing would be to ask your college seniors who have done their training from that company. You can then take a decision.
Hope this helps !
thanks for everything sir
Hi there ,
I am in 3rd year of B.pharma.
I am kinda confused about my career after this .. I feel blank .. I am interested in pharmD after this but I don’t know if it’s better than M.pharma.
Kindly suggest me all I need to know about Pharma D and scope of it and whether to opt for this or M.pharma .
Please guide me .
If you are interested in community pharmacy, hospital administration, healthcare management or public health, then Pharm.D. is definitely a better option.
Hello Sir,
Myself shivani I have done my B.pharma from indrapratha university and now I have got admission in both M.pharma in Pharamacology and Msc pharmacology from AIIMS. Now I am very much confused where to take final admission. I know about M pharma that it is more professional course compare to Msc in pharmacology but the college offering this course is very famous please guide me regarding the scope of both and which one is best to choose.
thankyou. Waiting for the reply.
Hi Shivani,
Be it industry or academia, the MSc Pharmacology degree from AIIMS will give you much better prospects. Only if the M.Pharm offer is from NIPER, you can give it a second thought. An MSc is in no way inferior to an M.Pharm. qualification. In fact, the MSc degree might give you better scopes for the research career.
Good Luck !
Hello sir,
I am pursueing dual integrated course in biotechnology (btech+mtech) , and is interested in biomedical sciences..!
Well, Thanku so much for the vedio on YouTube and your blog on biomedical sciences..! Has really helped me in understanding..!
Since , I am interested in medical and healthcare sort if field ,so My question is that since I am pursuing a 5years integrated course,So, can I directly apply for PhD in abroad universities (in USA, UK,Australia or Germany) .? Or would have to pursue masters again in this program..? And to become a biomedical scientist at industrial level , is it necessary to pursue PhD.?
Also, can u name /suggest some good universities in the countries in the field of biomedical sciences..?
It would be very kind and helpful by your reply..!
Thanking you..!
Hi Jhumi,
With a 5-year integrated Masters, you are definitely eligible for PhD in US, UK, Germany, Australia and other countries. You won’t need a second Masters degree.
I hope you have gone through one of my earlier articles (https://www.mbacrystalball.com/blog/2014/11/23/biomedical-science-jobs-careers/). A PhD degree will certainly give you a good boost for a research role in the industry. But, you can also get there with your current qualification.
Top universities in abroad for biomedical sciences – it would be a long list. I would suggest you to refer to QS (Top Universities) and THE subject rankings (life sciences).
All The Best !
goodevening Tanmoy sir , i am b.pharm graduate and want to know is m.pharm better or pharm.d as i wish to work abroad .
Hi Pia,
Both are equally good options in terms of getting a job in abroad. Please refer to my earlier post – https://www.careerizma.com/blog/pharmacy-careers-jobs/
hi sir,
I’m a Bpharm graduate. Interested in pharmD as well as clinical pharmacy.
If I do my pharmD from India and opt for Clinical pharmacy course in the USA, do the colleges in USA accept this?
Hi Neha,
After doing Pharm.D, you can certainly go for Masters in Clinical Pharmacy in the US (provided you clear GRE, MCAT or PCAT).
However, I would suggest to go for Masters in USA right after your B.Pharm (I assume going for Masters in the US is your next goal). Doing two Masters would be a waste of time; especially because Pharm.D after B.Pharm will take another 3 years to complete. Of course a Pharm.D (after B.Pharm) might give you some edge over other applicants. But, it might also create a negative impact on the admission officers – “Why the candidate wants to do a second Masters in a similar field?” or the US visa authorities might also reject your visa (if it is not a top tier university) by assuming that you are just going to study in the US for immigration purpose.
Feel free to ask in case you have further doubts !
Hi Sir,
I’m currently studying in second year MBBS. I want to persue my higher studies from abroad. I am inclined towards genetics and biomedical sciences more rather than going in for PG in clinical subjects. Just wanted to know whether there are good options for persuing biomedical sciences abroad after mbbs and is there any particular exam for it like there’s USMLE for PG after mbbs in the US.
He Mehak,
You can go for MSc in Medical / Biomedical Sciences after MBBS. You can also choose MSc in Genetics / Molecular Genetics.
If you want to do your MS from the US, you will need to appear for GRE (apart from IELTS or TOEFL). For other countries (UK, Canada, Ireland, Australia) you just need IELTS or TOEFL.
Hope this helps !
Your exhaustive and knowledge filled replies are a we inspiring Sir. I am currently a BTech Biotechnology pass out from VIT, Vellore, with a 9point CGPA throughout my academic career. I have acceptance from SUNY, Albany, for their MS, Biology Program, with MCDN concentration. I should have been starting this Fall, but my visa is in administrative processing. I also have acceptance from Lund University, Sweden, in MS, Biomolecular Genetics Program. If the US visa is denied, should I go for Sweden, if that is, I get back my passport in time? I have done badly in GATE BT-412 marks, AIR 910. Thinking I will go abroad, I missed all my CCMT counselling. Now I have Amity, MTech Biotechnology or VIT, MS, Biomedical Genetics options.
I am confused and depressed.
By the way, I did my BTech Final Semester project at Hedstrom Laboratory, Brandeis University, Waltham, on mTor pathways for Ubiquilin and am absolutely fascinated by the research ethic of the scientists in the US.
Please let me know what I should do. I am lost and broken.
Hi Modhurika,
SUNY Albany is decent enough; so I don’t think there should be any trouble with visa (provided other documentations are fine).
Lund University is an excellent university. In fact you can also choose that over SUNY Albany for Biomolecular Genetics. Lund is really prestigious and the research infrastructure is awesome. Lund comes within the Top 100 in the World; and in the Top 50 in the world for Life Sciences.
You have got admits from two good universities; in fact getting an admit from Lund is a great achievement. So, no need to be feel depressed or broken.